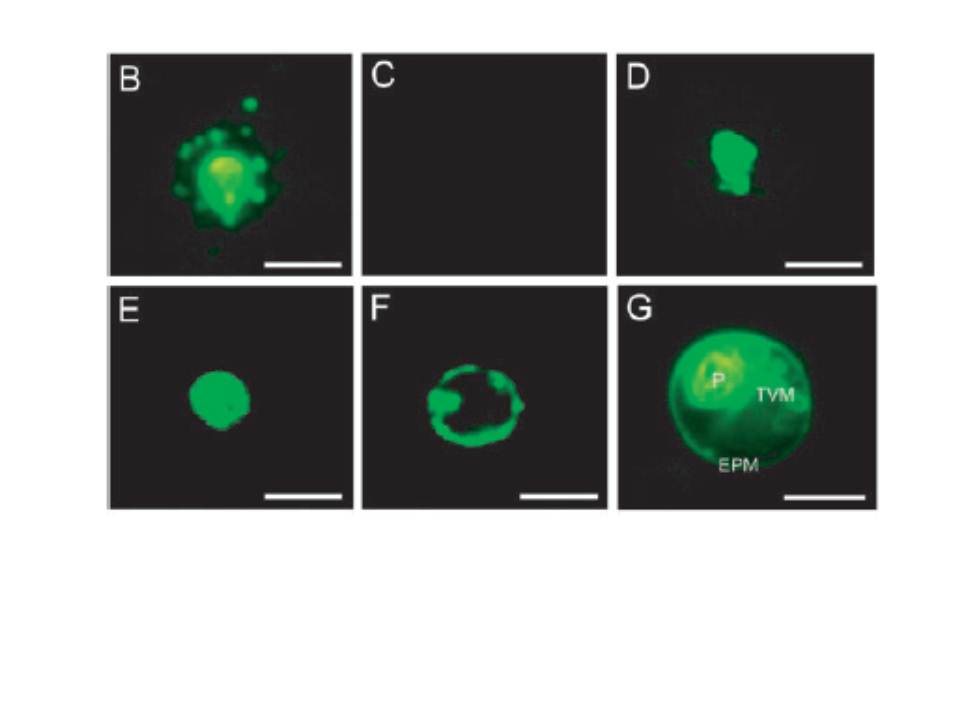
Immunohistochemical detection of PfNSF. (B), PfNSF in noninfected erythrocyte (C), vacuolar H1-ATPase subunit PF13_0065 A (V-ATPase) (D), H1-pumping pyrophosphatase (V-PPase) PF14_0541 (E), and the serine repeat antigen protein (F) in parasitized erythrocytes. The parasitized erythrocytes were immunostained with the indicated antibodies and then observed by fluorescence microscopy. Vital staining with C5-ceramide was also performed to reveal localization of the tubovesicular membrane networks (TVM) (G). P, P. falciparum cell; EMP, erythrocyte plasma membrane. Bar, 5 mm. the PfNSF immunoreactivity was present within the vesicular structures outside the parasite cells (B). No such extraparasitized vesicular structures were observed in the immunoreactivities against antibodies for vacuolar H+-ATPase (D), H+-pumping pyrophosphatase (E), serine repeat antigen protein, markers for the peripheral space between the parasitophorus vacuolar membranes, and the plasma membrane of the malaria parasite (F).
Hayashi M, Taniguchi S, Ishizuka Y, Kim HS, Wataya Y, Yamamoto A, Moriyama Y. A homologue of N-ethylmaleimide-sensitive factor in the malaria parasite Plasmodium falciparum is exported and localized in vesicular structures in the cytoplasm of infected erythrocytes in the brefeldin A-sensitive pathway. J Biol Chem. 2001 May 4;276(18):15249-55.
Other associated proteins
| PFID | Formal Annotation |
|---|---|
| PF3D7_0303000 | N-ethylmaleimide-sensitive fusion protein |
| PF3D7_1456800 | V-type H(+)-translocating pyrophosphatase, putative, VP1 |