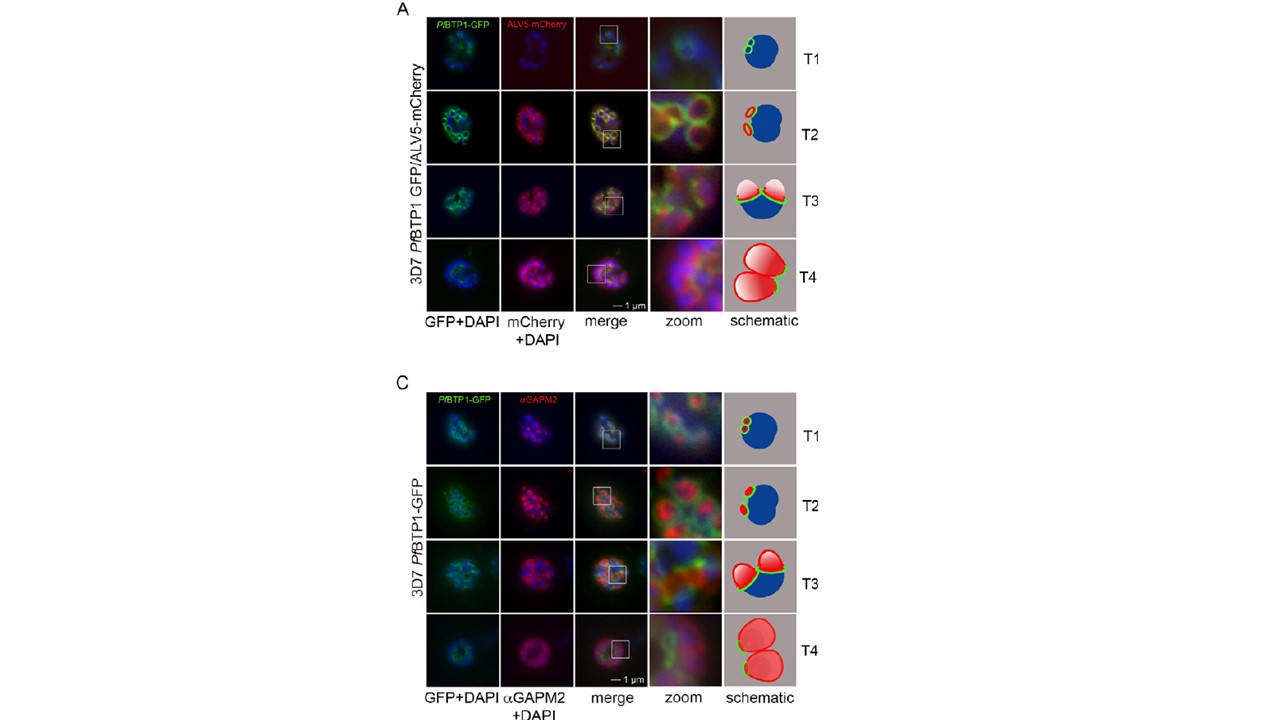
Colocalization of BTP1 with ALV5 and GAPM2 during inner membrane complex (IMC) biogenesis. (A) Colocalization of BTP1–GFP with ALV5–mCherry (red) in unfixed parasites. The initial ring structure marked by BTP1–GFP precedes the appearance of Alveolin 5 (ALV5) (T1). The emerging Alveolin-5-defined ring is associated but distinct from that marked by BTP1 (T2). During IMC expansion, BTP1 remains rim associated (T3) and, at the end of daughter cell formation, is concentrated at the basal end with ALV5 equally distributed within the IMC (T4). (C) Colocalization of BTP1–GFP with GAPM2 (red) in fixed parasites. Both proteins appeared at about the same time, marking the onset of IMC biogenesis (T1). GAPM2 stains small ring–shaped formations that are encircled by BTP1–GFP (T2). With elongation and propagation of the IMC from the apical towards the basal end of the forming daughter cell, GAPM2 is equally distributed within the forming IMC, but BTP1 is only present at the rim of the nascent IMC (T3) and, at the end of schizogony, is concentrated at the basal pole of the daughter cell (T4). Nuclei were stained with DAPI (blue). Enlargement of selected areas are marked with a white square and referred to as ‘zoom’ (a fourfold magnification). The bottom row shows a schematic representation of the spatio-temporal distribution of BTP1 and GAPM2.
Kono M, Heincke D, Wilcke L, Wong TW, Bruns C, Herrmann S, Spielmann T, Gilberger TW. Pellicle formation in the malaria parasite. J Cell Sci. 2016 129(4):673-80.
Other associated proteins
| PFID | Formal Annotation |
|---|---|
| PF3D7_0611600 | basal complex transmembrane protein 1 |
| PF3D7_1003600 | inner membrane complex protein 1c, putative |