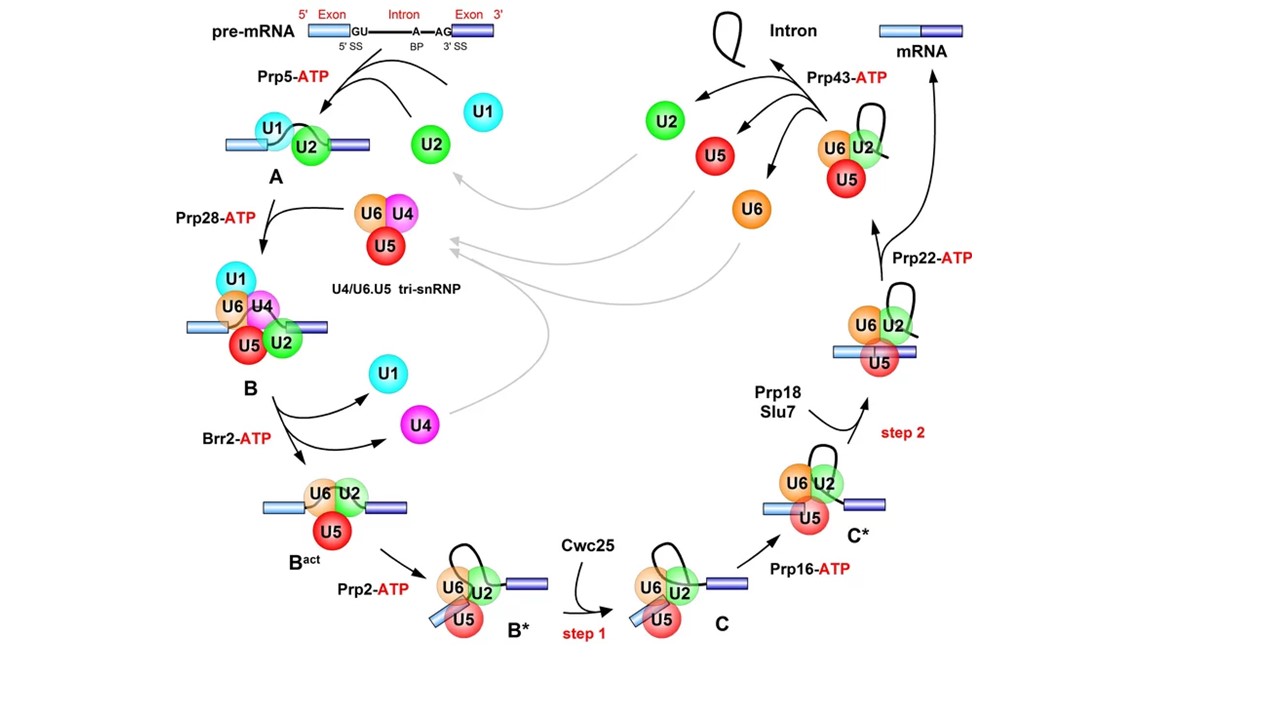
Assembly of the spliceosome by the stepwise binding of the snRNPs to the pre-mRNA. In the early phase of spliceosome assembly, the U1 snRNP binds to the 5' splice site (5' SS: where exon 1 ends and the intron begins), and the U2 snRNP binds to the so-called branch point (BP: near the 3' end of the intron). This spliceosome assembly intermediate is called the A complex. The subsequent binding of the U4/U6.U5 tri snRNP complex gives rise to the precatalytic B complex. The catalytic activation of the spliceosome takes place in two steps. In the first, the RNA helicase Brr2 acts to produce the Bact complex and in the second, the RNA helicase Prp2 facilitates the formation of the B* complex. This has a functional active site and, following the recruitment of the protein Cwc25, the first step of splicing takes place. In this step, the phosphodiester bond at the 5' splice site is cleaved and, at the same time, the 5' end of the intron becomes linked to the 2' hydroxyl group of an adenosine at the branch point. In the next step, the RNA helicase Prp16 converts the spliceosome to the C* complex, which – with the help of the proteins Prp18 and Slu7 – carries out the second catalytic step of the splicing reaction. In this step, the phosphodiester bond at the 3' splice site (3' SS: where the intron ends and exon 2 begins) is cleaved and at the same time the two exons are joined to one another. The intron is released from the spliceosomal complex in the form of a lasso (lariat) and the snRNPs are recycled for subsequent rounds of splicing. The dissociation phase of the spliceosome requires catalysis by the RNA helicases Prp22 and 43. "ATP" indicates the steps that require ATP molecules as a source of chemical energy.
PubMed Article: Pulmonary trophoblastic deportation. Cytologic aspects