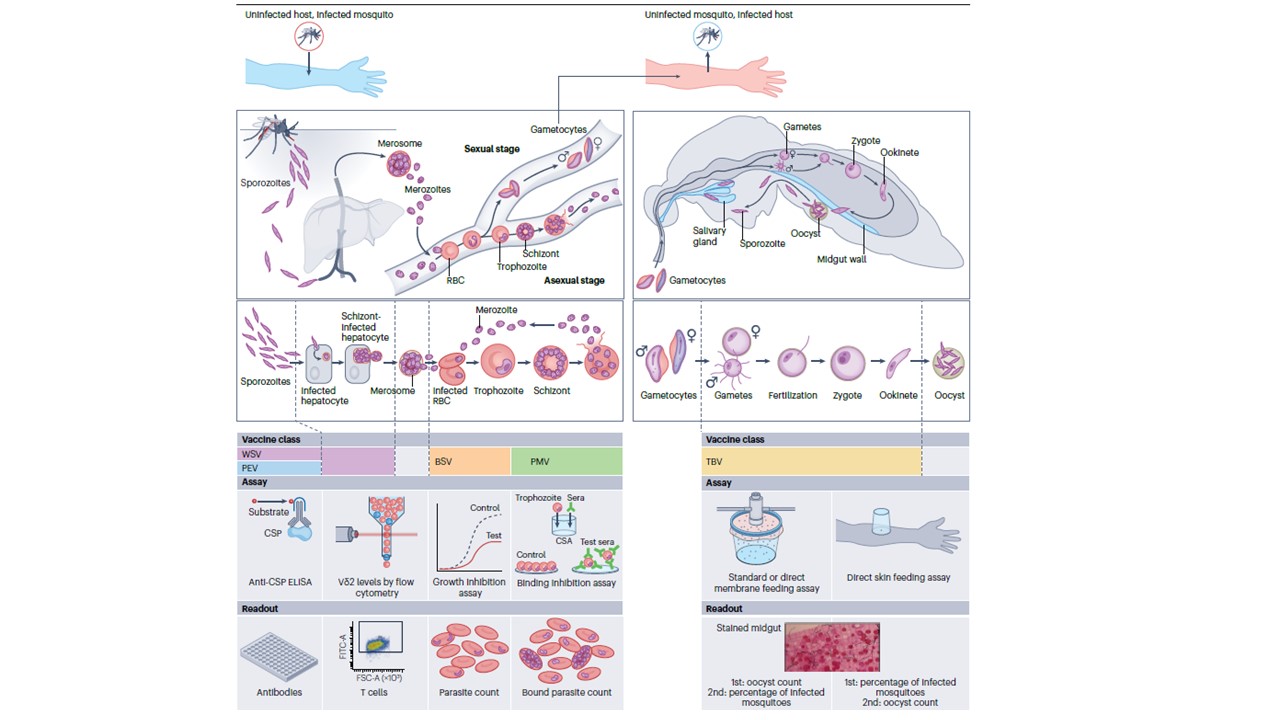
Malaria vaccine classes targeting different stages of the parasite life cycle with associated bioassays. The Plasmodium parasite life cycle stages are presented as a framework to understand different malaria approaches, starting with inoculation of sporozoites by an infected mosquito into an uninfected person. Parasites pass through pre-erythrocytic (sporozoite and the liver) stages, blood stages and mosquito stages. Below each life cycle stage, parasite forms targeted by different vaccine classes are indicated by coloured bars.
For each vaccine class, the associated bioassays and readouts used to measure vaccine activity or surrogate efficacy are presented. Although measurements of some antibody or cellular responses have been associated with efficacy in individual trials, no clinically validated surrogate endpoint of efficacy exists for any malaria vaccine (see the section ‘Understanding immune responses and correlates of protection’. Pre-erythrocytic vaccine (PEV) efficacy has been associated with circumsporozoite protein (CSP) ELISA titre and, for whole-sporozoite vaccines (WSV), with levels of Vδ2 γδ T cells. Functional activity of blood-stage vaccines (BSV) is often measured in growth inhibition assays, which test the ability of serum or IgG to reduce parasite invasion or multiplication during in vitro culture. The functional activity of placental malaria vaccine (PMV) candidates is quantified in binding-inhibition assays, which test serum or IgG for inhibition of parasite binding to chondroitin sulfate A (CSA). Transmission-blocking vaccines (TBVs) are assessed by in vitro and in vivo assays; standard or direct membrane feeding assays wherein antisera are added to in vitro cultured gametocytes (for standard membrane feeding assays) or trial participant blood (for direct membrane feeding assays) and fed to insectary-raised mosquitoes to assess transmission-blocking activity; and in vivo direct skin feeding assays, wherein mosquitoes feed directly on skin of participants to assess transmission of malaria parasites. Direct skin feeding, which recapitulates human-to-mosquito parasite transmission as it naturally occurs, measures vaccine effects (for example, reduction in percentage of infected mosquitoes) that are appropriate for mathematical modelling and can serve as reasonably likely surrogate efficacy endpoints for accelerated or conditional
licensure. RBC, red blood cell. Duffy PE, Gorres JP, Healy SA, Fried M. Malaria vaccines: a new era of prevention and control. Nat Rev Microbiol. 2024 Jul 18. doi: 10.1038/s41579-024-01065-7.