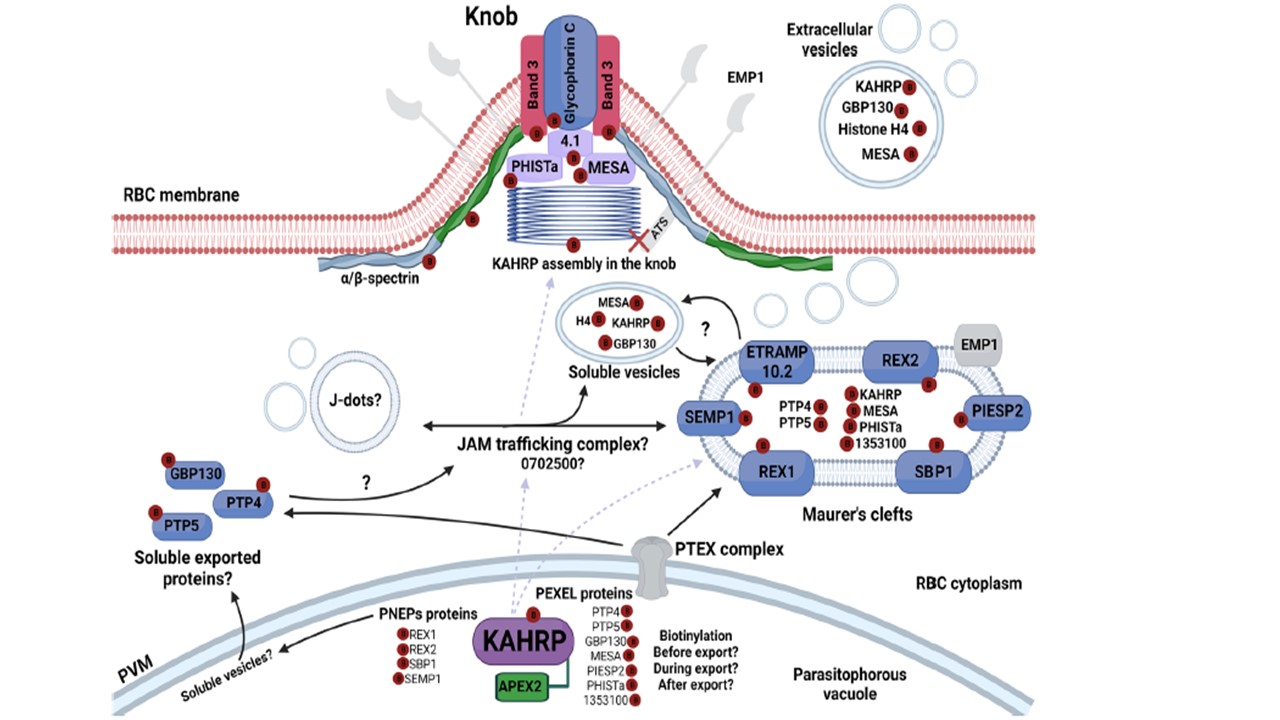
Diagram of potential cellular locations for interaction between KAHRP and parasite and host cell partners during protein export. Based on our proteomic data, APEX2-tagged KAHRP has biotinylated a set of proteins. Different trafficking routes throughout iRBC, including translocation via the PTEX complex and possibly alternative export pathways (e.g. PNEP proteins) are proposed to be used by KAHRP until its destination at knobs under the RBC membrane. Identified biotinylated proteins are labelled by a red circle with the letter B. PfEMP1, the main partner of KAHRP is described as a periphery protein found in the MC and its ATS domain does not interact with KAHRP. These topological features probably hampered APEX2 biotinylation. Other MC proteins, such as SBP1, PIESP2, REX1, REX2, SEMP1 and ETRAMP10.2 were found biotinylated in this study in agreement with the literature. Moreover, host proteins such as α/β-spectrin, protein 4.1, glycophorin and band 3 that compose cytoskeleton erythrocyte proteins were also biotinylated by KAHRPAPEX2, confirming the involvement of these proteins on the knob architecture. It is known that MC and J-dots comprise the JAM trafficking complex. One protein from our dataset found in this trafficking hub connecting these structures is the Plasmodium exported protein (PF3D7_0702500). Of special note, KAHRP and other proteins, such as GBP130, histone H4 and MESA, which were also found in our dataset as biotinylated, were reported to be in extracellular vesicles from ring-stage parasites, suggesting that protein export mechanisms in P. falciparum is much more complex than presumed. Charneau S, de Oliveira LS, Zenonos Z, Hopp CS, Bastos IMD, Loew D, Lombard B, Pandolfo Silveira A, de Carvalho Nardeli Basílio Lobo G, Bao SN, Grellier P, Rayner JC. APEX2-based proximity proteomic analysis identifies candidate interactors for Plasmodium falciparum knob-associated histidine-rich protein in infected erythrocytes. Sci Rep. 2024 14(1):11242. PMID: 38755230