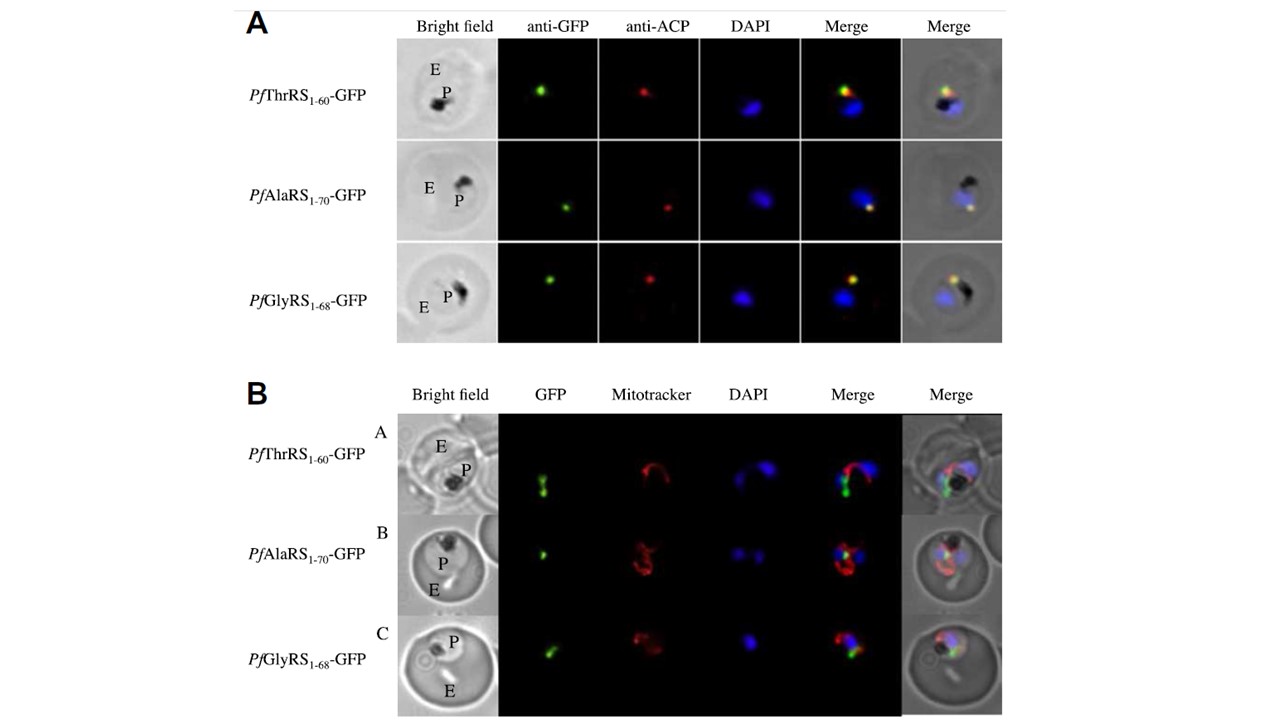
The N-terminus of Plasmodium falciparum (Pf)ThrRS, PfGlyRS and PfAlaRS directs apicoplast targeting. (A) PfThrRS1–60-GFP, PfAlaRS1–70-GFP and PfGlyRS1–68-GFP transfected parasites were labelled with anti-GFP and anti-acyl carrier protein antibodies and DAPI. The aminoacyl-tRNA synthetase–GFP fusions overlap with the apicoplast marker ACP, demonstrating that the N-terminus of these gene products is sufficient for apicoplast targeting. Bright field panels show the parasite (P) residing in its host erythrocyte (E). (B) Live cell microscopy shows that the N-terminal fragments of the aaRSs do not direct trafficking to mitochondria. Live parasites were stained using Mitotracker to highlight the mitochondria and this was compared with the GFP signal for each of the aaRS–GFP parasites. In all cases the mitochondria labelled a distinct and
separate organelle from the GFP, indicating that these N-terminal leaders do not direct mitochondrial trafficking. (C) Western blotting (using anti-GFP antibody) of parasites transfected with the N-termini of the aaRS proteins fused to GFP shows that the apicoplast targeting leader is cleaved as expected. In each of the N-terminal aaRS–GFP fusions a doublet is seen corresponding to a longer version retaining the transit peptide (but lacking the co-translationally-removed signal peptide) and a shorter version with the transit peptide removed. A similar pattern is frequently observed with apicoplast-targeted proteins.
Jackson KE, Pham JS, Kwek M, De Silva NS, Allen SM, Goodman CD, McFadden GI, Ribas de Pouplana L, Ralph SA. Dual targeting of aminoacyl-tRNA synthetases to the apicoplast and cytosol in Plasmodium falciparum. Int J Parasitol. 2012 42(2):177-86. PMID: 22222968.
Other associated proteins
| PFID | Formal Annotation |
|---|---|
| PF3D7_1367700 | alanine--tRNA ligase |
| PF3D7_1420400 | glycine--tRNA ligase |