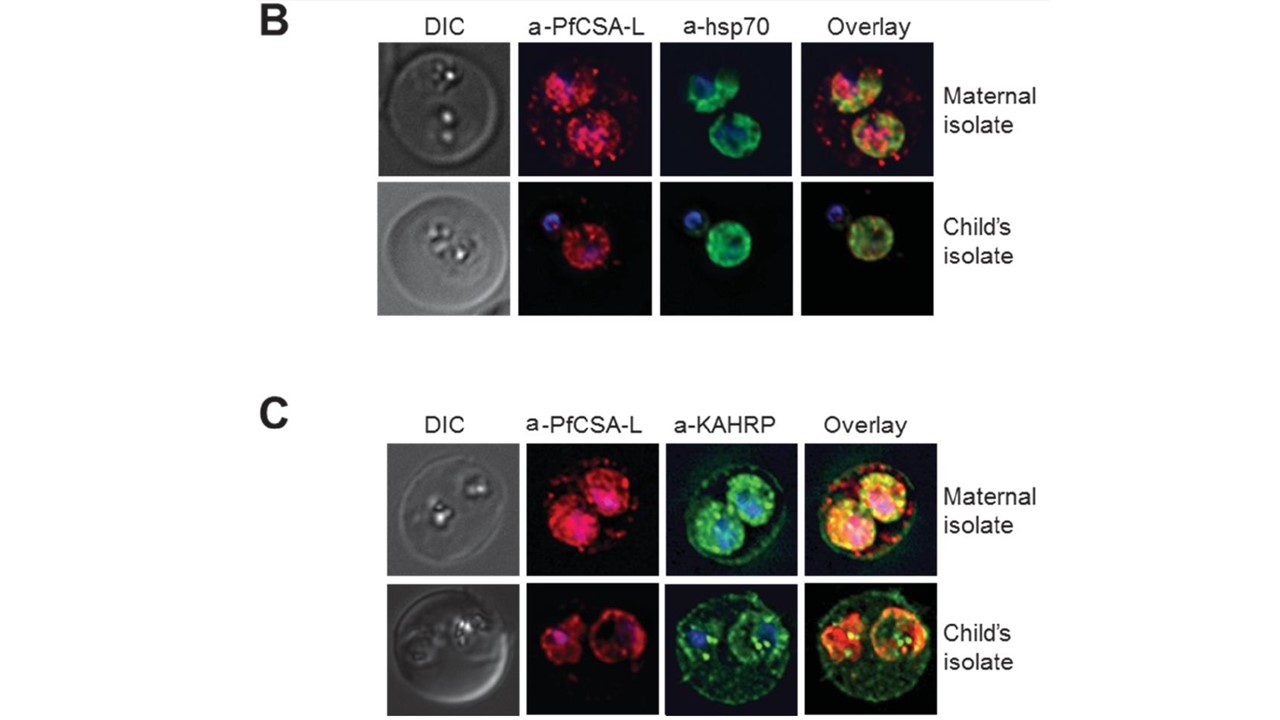
PfCSA-L is membrane-associated and expressed on the surface and interior of maternal iRBC. (B) Immunofluorescence of fixed, permeabilized maternal and children’s iRBC with rabbit PfCSA-L-specific antibodies (red) and anti-HSP70 monoclonal antibodies (green); first panel and last panel represent Differential Interference Contrast (DIC) and overlay images respectively. Images are representative of 3 assays with each isolate. (C) Immunoflourescence of fixed, permeabilized maternal and children’s iRBC with rabbit PfCSA-L-specific antibodies (red) and anti-KAHRP monoclonal antibody (green); first and last panel represent DIC and overlay images respectively. Images are representative of 3 assays with each isolate. Both VAR2CSA and PfCSA-L are knob surface proteins anchored to the iRBC membrane by protein-protein interactions rather than exclusively through protein-lipid interactions. VAR2CSA behaves as an integral membrane protein, and peripheral membrane protein. These two proteins not only co-localize on the surface of the iRBC, but are proximal to each other, and capable of directly interacting with nanomolar affinity. We suggest that the majority of membrane-associated PfCSA-L is exposed on the iRBC surface and anchored as a peripheral membrane protein in complex with VAR2CSA.
Keitany GJ, Jenkins BJ, Obiakor HT, Daniel S, Muehlenbachs A, Semblat JP, Gamain B, Doritchamou JYA, Desai SA, MacDonald NJ, Narum DL, Morrison R, Saveria T, Vignali M, Oleinikov AV, Fried M, Duffy PE. An invariant protein that co-localizes with VAR2CSA on Plasmodium falciparum-infected red cells binds to chondroitin sulfate A. J Infect Dis. 2021 Oct 29:jiab550
Other associated proteins
| PFID | Formal Annotation |
|---|---|
| PF3D7_0202000 | knob-associated histidine-rich protein |
| PF3D7_0818900 | PfHsp70-1 |
| PF3D7_1001000 | chondroitin sulfate a ligand; plasmodium exported protein (hyp12) |