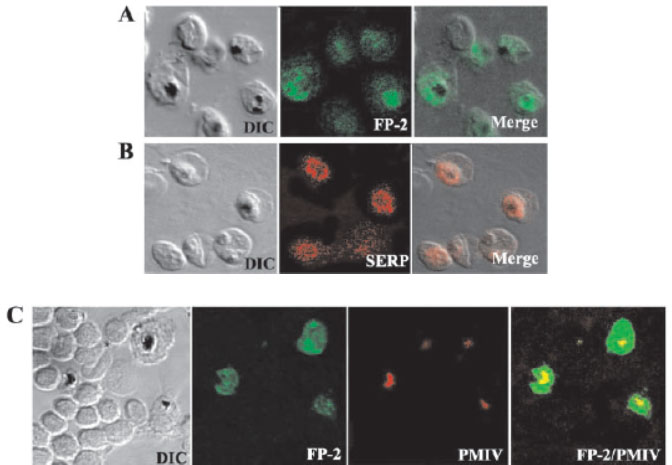
Immunolocalization of FP-2 by confocal microscopy. Erythrocytes infected with P. falciparum trophozoites were smeared on a glass slide, fixed with ice-cold methanol, and incubated with polyclonal FP-2 (A) or SERP (B) antibodies followed by Alexa 488 (green) or Alexa 594 (red) conjugated goat anti-rabbit IgG. Merged images of differential interference contrast (DIC) and fluorescence microscopy are shown on the right. C and D, slides were incubated with polyclonal FP-2 together with either plasmepsin IV (C) antibodies followed by Alexa-488-conjugated (green) goat anti-rabbit IgG and Alexa 594-conjugated (red) goat anti-mouse IgG. Merged images of green and red micrographs are shown on the right. The FP-2 antibody stained the entire trophozoite and vesicles or vesicle-like structures of unknown identity were seen extending into the erythrocyte cytoplasm. Co-staining of infected erythrocytes with antibodies to plasmepsin IV, which is present exclusively in the food, confirmed that FP-2 is present in the food vacuole as well as in the area that lies outside of the food vacuole (C). A comparison of FP-2 and SERP staining shows that the FP-2 is localized in the PVM as well as in a compartment that is distinct from the PVM.
Dhawan S, Dua M, Chishti AH, Hanspal M. Ankyrin peptide blocks falcipain-2-mediated malaria parasite release from red blood cells. J Biol Chem. 2003 278:30180-6
Other associated proteins
| PFID | Formal Annotation |
|---|---|
| PF3D7_0219400 | ribosome associated membrane protein RAMP4, putative |
| PF3D7_1115700 | cysteine proteinase falcipain 2a falcipain 2 |