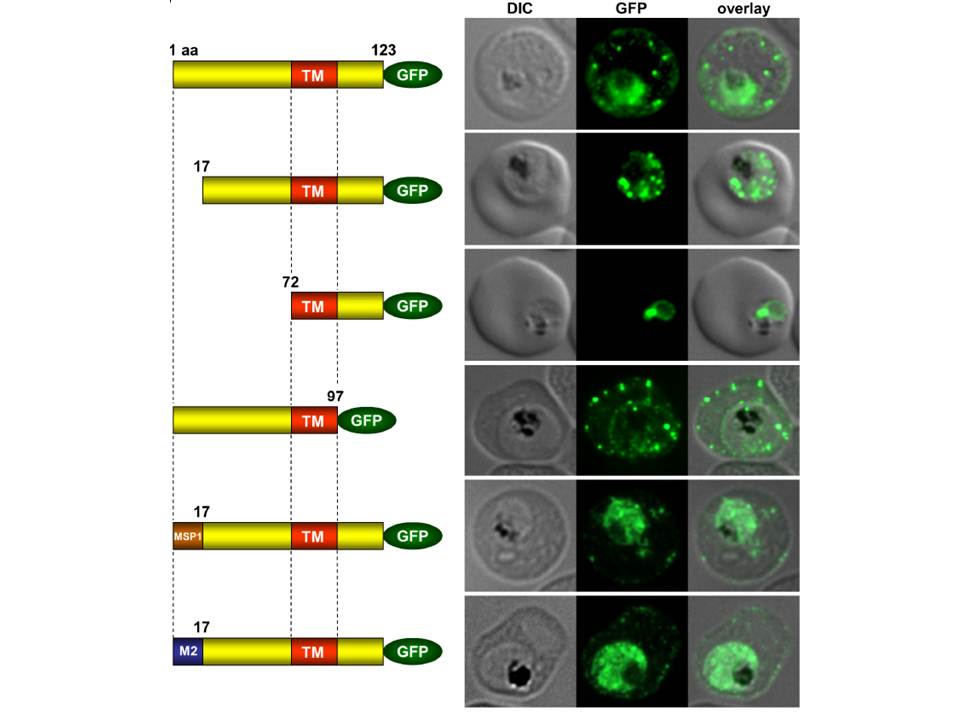
Immunofluorescence assays of MeOH-fixed RBCs infected with SEMP1-KO parasites expressing full-length and truncated or mutated forms of SEMP1 C-terminally fused to GFP. Expressed SEMP1 was labelled with rabbit a-GFP antibodies. The transmembrane domain is depicted in red (TM), a MSP1 signal peptide in brown (MSP1) and the MAHRP2 N-terminus in blue (M2). deletion of the complete C-terminus of SEMP1 (SEMP11–97-GFP) did not impair export and resulted in correct localisation whilst deletion of the first 16 amino acids (SEMP117–123-GFP) of the N-terminus resulted in an impaired export from the parasite. The deletion of 71 amino acids (SEMP172–123-GFP) of the N-terminus prevented export and seemed to have resulted in concentration of SEMP1 in or at the endoplasmic reticulum. If the first 16 amino acids of SEMP1 were replaced by the corresponding amino acids of MAHRP2 (M21–16SEMP117–123-GFP) or the classical signal peptide of MSP1 (MSP11–16SEMP117–123-GFP) the fusion protein was exported in both cases albeit with lower efficiency.
Dietz O, Rusch S, Brand F, Mundwiler-Pachlatko E, Gaida A, Voss T, Beck HP. Characterization of the Small Exported Plasmodium falciparum Membrane Protein SEMP1. PLoS One. 2014 9(7):e103272.
Other associated proteins
| PFID | Formal Annotation |
|---|---|
| PF3D7_0702400 | small exported membrane protein 1 |
| PF3D7_1370300 | membrane associated histidine-rich protein |