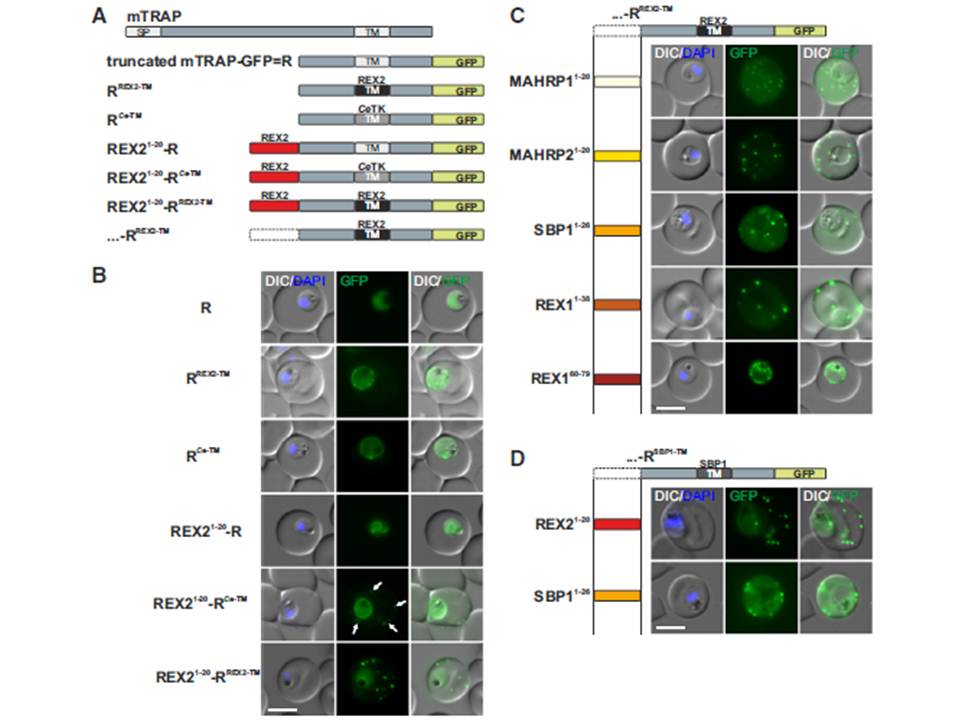
N Termini of PNEPs Are Sufficient for Export of RREX2-TM, (A) Schematic of mTRAP fusion constructs. (B) Representative images of live P. falciparum parasites expressing the constructs shown in (A). DIC, differential interference contrast; nuclei were stained with DAPI. Arrows indicate limited staining reminiscent of Maurer’s clefts. (C and D) Images of live P. falciparum parasites expressing RREX2-TM (C) and RSBP1-TM (D) fused with the PNEP N termini indicated. Panels are as in (B). Size bars represent 5 mm. The mTRAP backbone, different TMs, and the SP (signal peptide) are shown in different shades of gray; N-terminally appended regions of PNEPs are shown in shades from yellow to dark red. Ce-TM, C. elegans TK TM. The unmodified truncated mTRAP fused to GFP (‘‘R’’ for ‘‘reporter’’) does not contain any export-relevant sequences. This protein was found evenly distributed in the parasite cytosol (B). When the mTRAP TM in R was replaced with that of the PNEP REX2 or a previously used heterologous TM of a C.elegans tyrosine kinase (TK) (a TM that does not promote export of REX2 and does not change topology;), the R then entered the secretory pathway and was found in the parasite periphery (PPM, PV, or PVM) but was not exported.
Grüring C, Heiber A, Kruse F, Flemming S, Franci G, Colombo SF, Fasana E,
Schoeler H, Borgese N, Stunnenberg HG, Przyborski JM, Gilberger TW, Spielmann T. Uncovering common principles in protein export of malaria parasites. Cell Host Microbe. 2012 12(5):717-29.
Other associated proteins
| PFID | Formal Annotation |
|---|---|
| PF3D7_0936000 | ring-exported protein 2 |
| PF3D7_1353200 | membrane associated histidine-rich protein 2 |