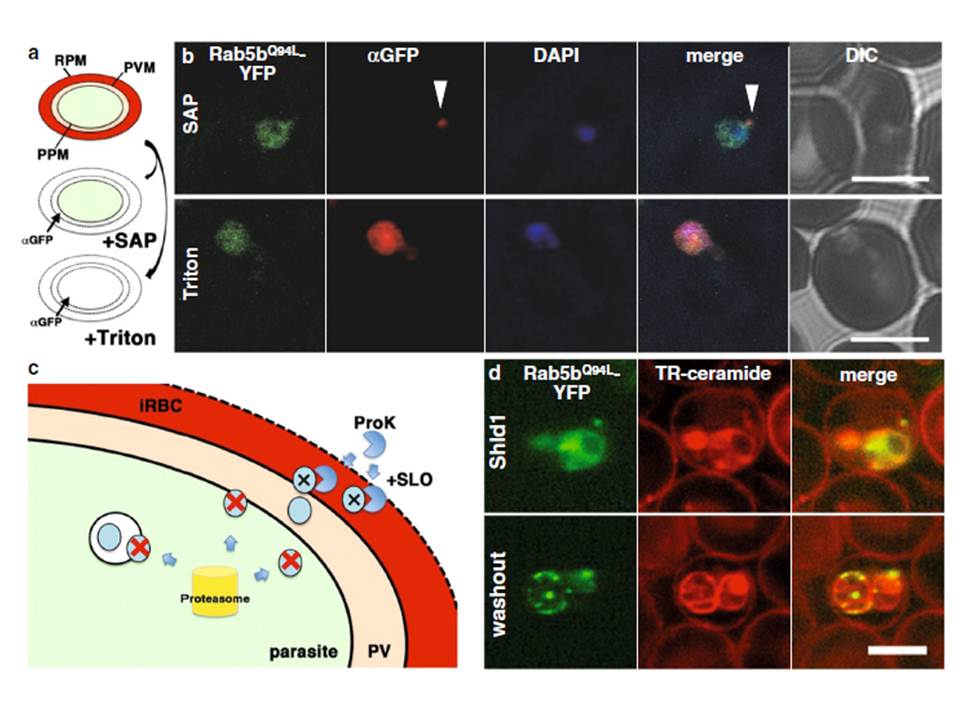
Transport of PfRab5bQ94L-YFP-DD to the cytoplasmic face of infected red blood cells on the TVN. a The selective permeabilization scheme. Saponin permeabilizes RBC plasma membrane (RPM) and PVM, which allows the detection of proteins localized to the outside of the parasite plasma membrane (PPM) or both sides of the PVM. Triton X-100 permeabilizes RPM, PVM, and PPM, allowing staining of the iRBCs, PV, and parasite cytosol with antibody. b Cells expressing PfRab5bQ94L-YFP-DD (green) were subjected to an antibody accessibility assay with anti-GFP antibody (red) prior to permeabilization with saponin (upper panel, SAP) or Triton X-100 (lower panel, Triton). After the saponin treatment, anti-GFP antibody labelled PfRab5bQ94L-YFP-DD secreted to the TVN (arrowhead), whereas PfRab5bQ94L-YFP-DD in the parasite cytosol was not labelled. Permeabilization with Triton X-100 allowed labelling of both the parasite cytosolic and TVN-localized PfRab5bQ94L-YFP-DD with anti-GFP antibody. c Schematic of the Shld1 washout assay and the protease accessibility assay. After removal of Shld1, cytosolic DD-tagged proteins are degraded (red cross and blue circle) by the parasite proteasome (yellow cylinder). DD-tagged proteins, inside of intracellular organelles or transported to the outside of the parasite (blue circular), are resistant to the proteasomal degradation. Since streptolysin O (SLO) permeabilizes RPM but not PVM, DD-tagged proteins in the RBC cytoplasm were selectively degraded by extracellular proteinase K (ProK) after permeabilization with SLO (black cross and blue circle). d Sub-cellular localization of PfRab5bQ94L-YFP-DD (green) after Shld1 stabilization for 24 h (upper panel, Shld1) and 2 h after Shld1 washout (lower panel, washout). After the removal of Shld1, the punctate signal of PfRab5bQ94L-YFP-DD localized to the TR-ceramide (red)-labelled parasite periphery. Bars 5 μm.
Ebine K, Hirai M, Sakaguchi M, Yahata K, Kaneko O, Saito-Nakano Y. Plasmodium Rab5b is secreted to the cytoplasmic face of the tubovesicular network in
infected red blood cells together with N-acylated adenylate kinase 2. Malar J. 2016 15:323.