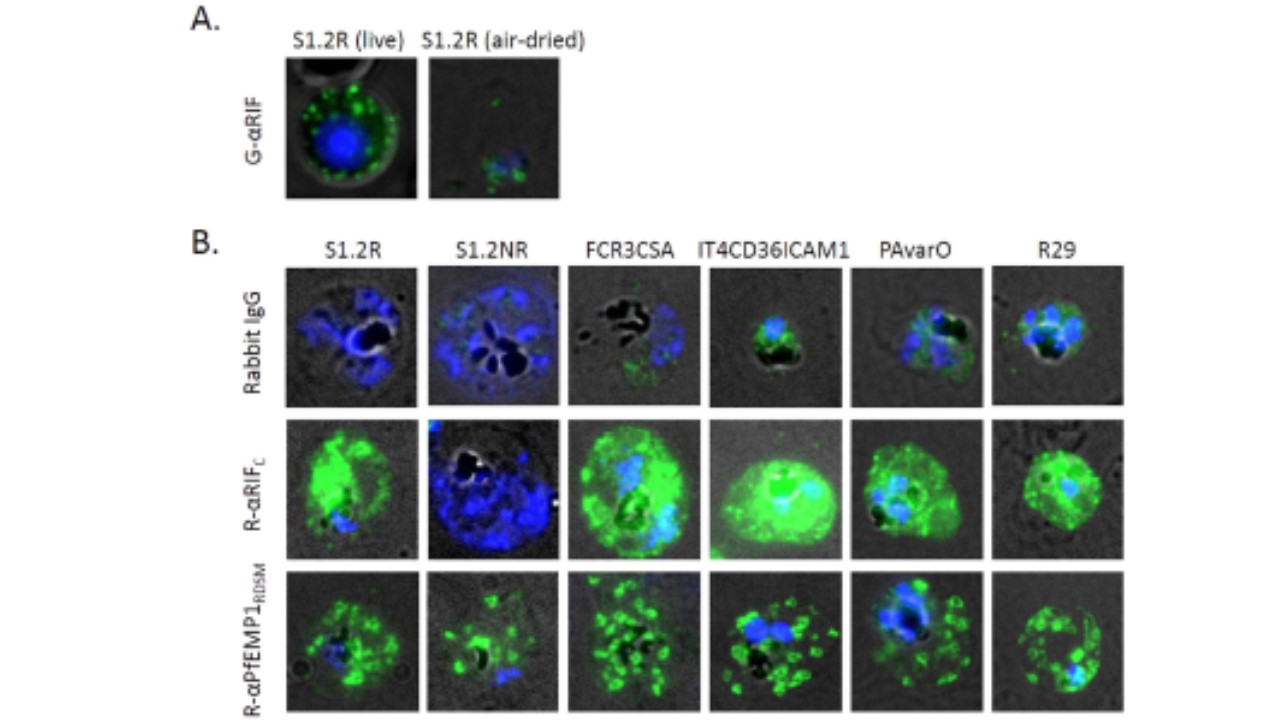
Indirect immunofluorescence micrographs. (A) Gα RIF IgG labelling of RIFINs on a live S1.2Rinfected erythrocyte and an air-dried iRBC. Unlike the antibody labeling of live S1.2R-infected cells which showed punctate surface-specific staining (left panel), air dried monolayers stained with Gα RIF IgG showed only a faint parasite cytoplasmic staining (right panel) suggesting that the native conformational (3D) epitope may have been lost during the fixing by desiccation. (B) Air-dried monolayers of erythrocytes infected with S1.2R, S1.2NR, FCR3CSA, IT4CD36ICAM1, PAvarO and R29 parasites stained with non-immune rabbit IgG (10 μ g/ml), anti-RIFIN Rα RIFC rabbit IgG (10 μg/ml) and Maurer’s cleft labelling by R-α PfEMP1RDSM IgG (8 μ g/ml). Alexa488-conjugated secondary anti-rabbit or anti-goat antibodies were used (1:200) as well as Vectashield with DAPI (nuclear staining). Merged channels of transmission light (black and white), green (Alexa 488) and blue (DAPI) and representative cells are shown here. Since desiccation results in breaks in the membrane and exposes the inner surface of the RBC membrane where the conserved RIFIN C-terminus is located, Rα RIFC IgG could readily label S1.2R-infected RBCs in a patchy manner. In comparison, IgG from Rα RIFI, R5α RIFC and R6α RIFC showed no notable staining (data not shown). Rα RIFC IgG was then applied to air-dried monolayers of other parasite strains to detect and localize potential A-RIFINs. While S1.2NR (Fig. 2B), NF54CSA and 3D7CD36ICAM1 (data not shown) had no notable staining, FCR3CSA, IT4CD36ICAM1, PAvarO and R29 parasites showed staining similar to S1.2 R.
Ch'ng JH, Sirel M, Zandian A, Del Pilar Quintana M, Chun Leung Chan S, Moll K, Tellgren-Roth A, Nilsson I, Nilsson P, Qundos U, Wahlgren M. Epitopes of anti-RIFIN antibodies and characterization of rif-expressing Plasmodium falciparum parasites by RNA sequencing. Sci Rep. 2017 7:43190.
Other associated proteins
| PFID | Formal Annotation |
|---|---|
| PfEMP1 | PfEMP1 |
| RIFIN | RIFIN |