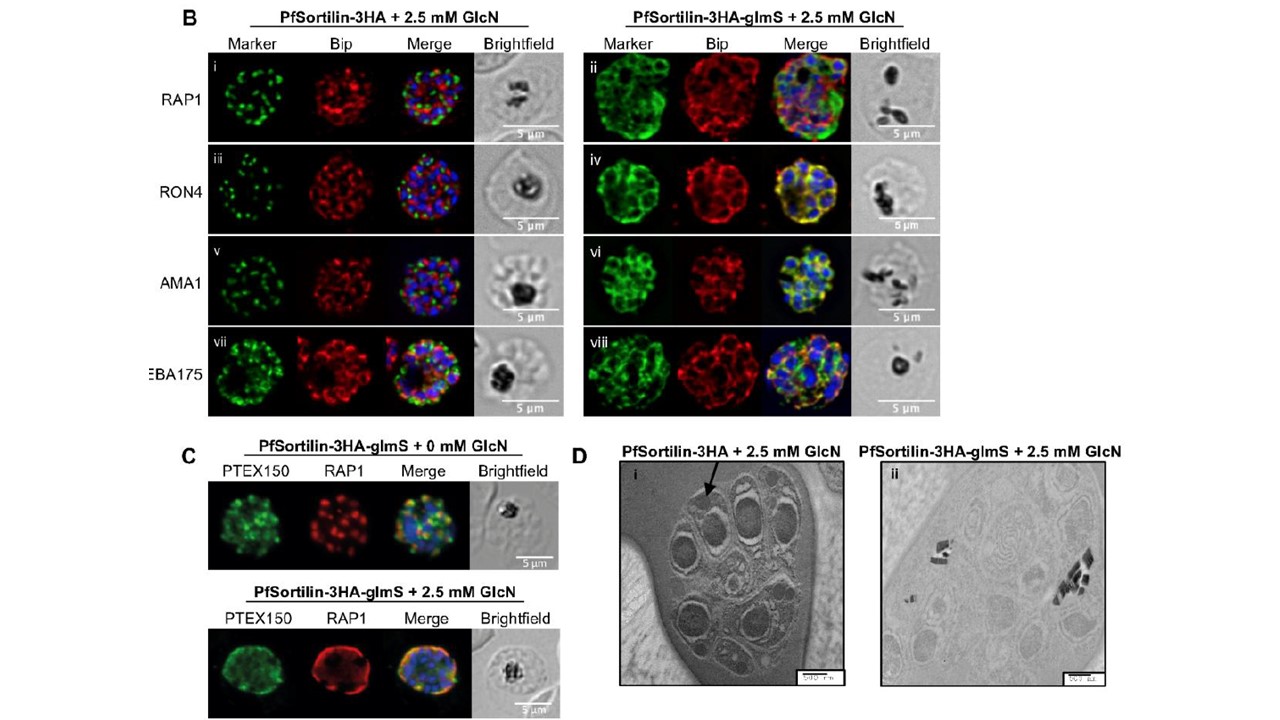
Knocking down PfSortilin abrogates trafficking to the apical organelles and prevents rhoptry formation. Colocalization with the ER marker Bip confirms that the apical proteins are trapped in the ER. (C) The dense granule protein PTEX150 is trafficked to the PV, like RAP1. (D) Electron microscopy reveals that rhoptries are not formed in absence of PfSortilin-3HA whilst they are easily seen in the control line (i, arrow). The marker label refers to the antibody used. Scale bars represent 5 μm for fluorescence images and 500 nm for electron microscopy. the fluorescence signal for the rhoptry bulb marker RAP1 in the KD line was found in the PV ,the default destination for soluble proteins entering the secretory pathway, and also sometimes around the DAPI-stained nuclei, reminiscent of the ER and this was confirmed by colocalization with Bip, an ER-resident protein (B).
Hallée S, Counihan NA, Matthews K, de Koning-Ward TF, Richard D. The malaria parasite Plasmodium falciparum Sortilin is essential for merozoite formation and apical complex biogenesis. Cell Microbiol. 2018 Mar 26:e12844 [Epub ahead of print]
Other associated proteins
| PFID | Formal Annotation |
|---|---|
| PF3D7_0731500 | erythrocyte binding antigen-175 |
| PF3D7_1116000 | rhoptry neck protein 4 |
| PF3D7_1133400 | apical membrane antigen 1 |
| PF3D7_1410400 | rhoptry-associated protein 1 |
| PF3D7_1451800 | sortilin |