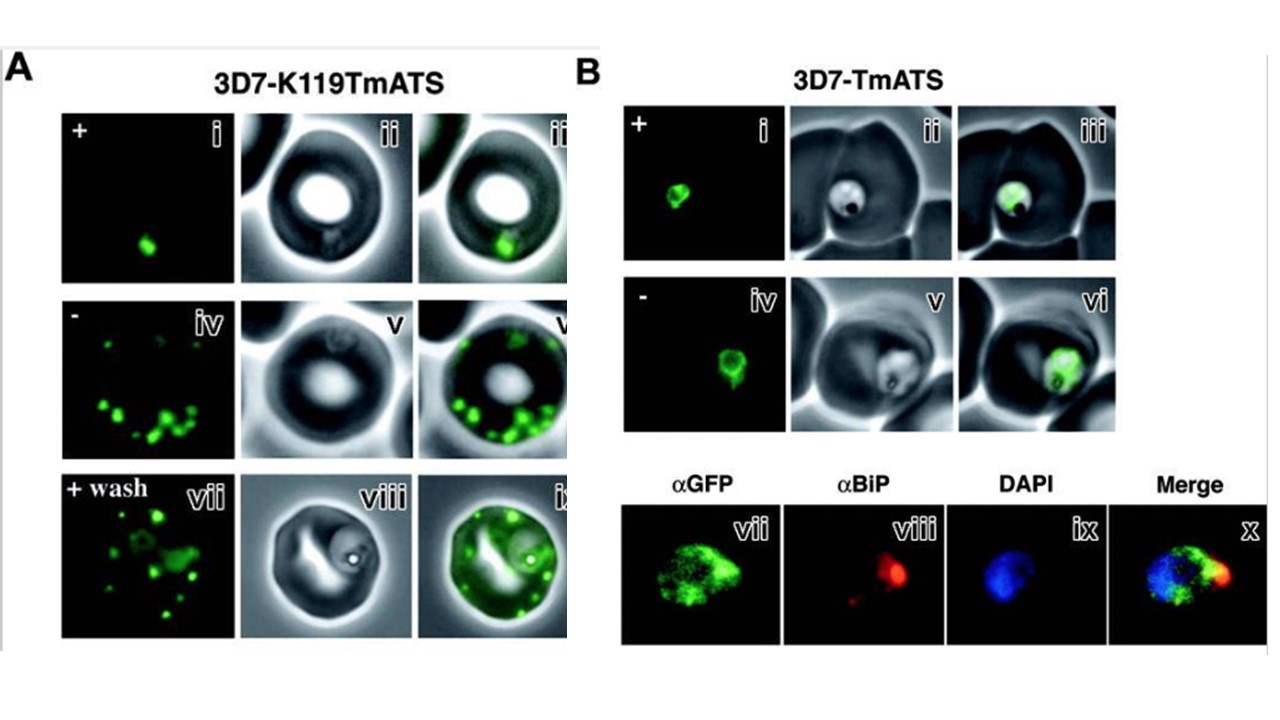
Trafficking of the K119TmATS-GFP protein is brefeldin A (BFA) sensitive. (Ai-ix) Live cells. GFP fluorescence (i,iv,vii); bright-field image (ii,v,viii); overlay (iii,vi,ix). Normal trafficking of the K119TmATS-GFP chimera was observed in the absence of BFA (-) or after washing, whereas BFA (+) treatment inhibited trafficking. (Ax-xiii) Immunofluorescence microscopy of fixed BFA-treated cells. (x) αGFP; (xi) αBiP; (xii) DAPI; (xiii) merge of subpanels x-xii. The chimera colocalizes with the ER marker BiP after incubation with BFA. (B) Brefeldin A treatment of 3D7-TmATS transfectants. (Bi-vi) Live cells in the presence (+) and absence (-) of BFA. GFP fluorescence (i,iv); phase-contrast images (ii,v); overlays (iii,vi). (Bvii-x) Immunofluorescence microscopy of fixed BFA-treated cells. (vii) αGFP antibody; (viii) αBiP antibody; (ix) DAPI; (x) merge of subpanels vii-ix. Trafficking of K119TmATS-GFP to the parasite membrane and beyond was inhibited by BFA (A). In BFA-treated cells a focus of fluorescence was observed near the periphery of the parasite with a diffuse pattern surrounding it. Removal of BFA allowed normal growth and trafficking of K119TmATS-GFP. We examined whether K119TmATS-GFP colocalized with the ER luminal protein PfBiP29 in BFA-treated cells (Ax-xiii).
Knuepfer E, Rug M, Klonis N, Tilley L, Cowman AF. Trafficking of the major virulence factor to the surface of transfected P. falciparum-infected erythrocytes. Blood. 2005 105(10):4078-87.
Other associated proteins
| PFID | Formal Annotation |
|---|---|
| PfEMP1 | PfEMP1 |