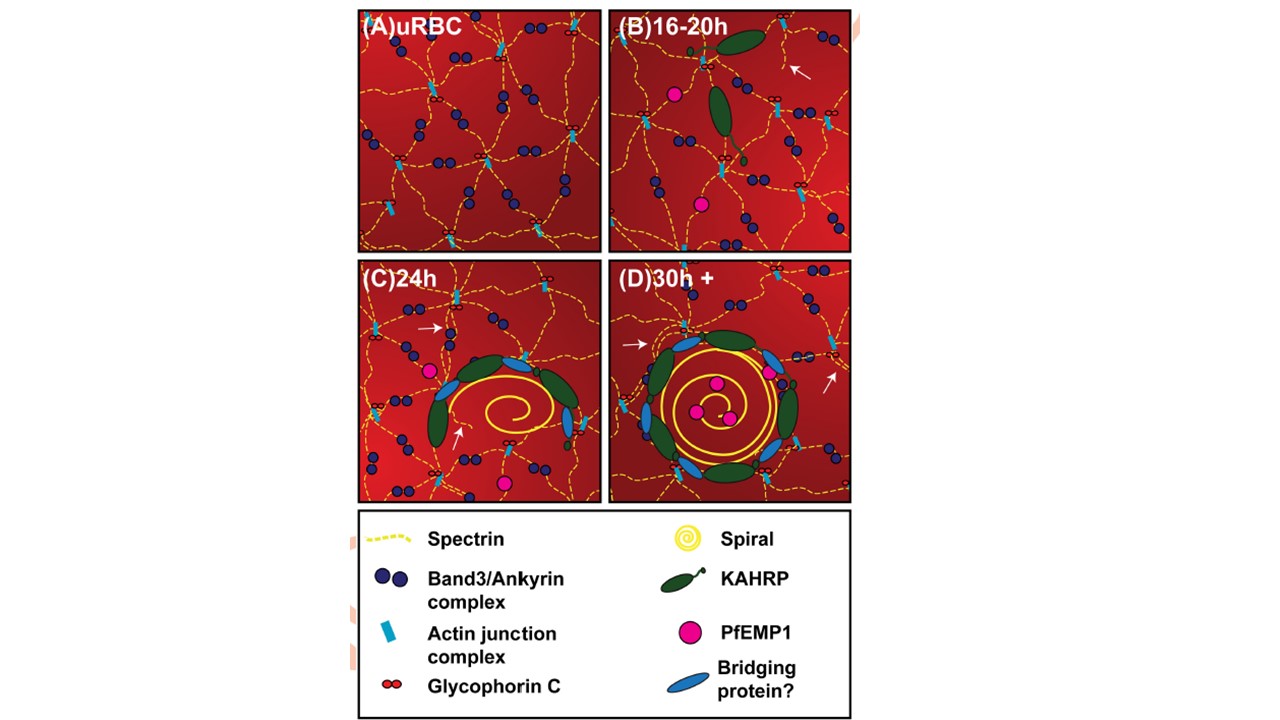
A model for virulence complex formation. A schematic diagram of the proposed model for assembly of the virulence complex. (A) The membrane skeleton of an uninfected RBC (uRBC). (B) KAHRP is delivered to the RBC membrane skeleton where it interacts with spectrin. Host cell remodeling is initiated (white arrows) via the dissociation of the actin junction points, allowing spectrin remodeling. PfEMP1 is delivered to the RBC membrane. (C) Host cell remodeling continues with spectrin octamers being formed (white arrow). KAHRP undergoes higher order assembly (with potential involvement of a bridging protein) forming a horseshoe-shaped structure that scaffolds the spiral knob core. PfEMP1 moves laterally to associate with the virulence complex. (D) KAHRP modules form the base of the knob complex. Most of the PfEMP1 is assembled adjacent to or at knob complexes.
Looker O, Blanch AJ, Liu B, Nunez-Iglesias J, McMillan PJ, Tilley L, Dixon MWA. The knob protein KAHRP assembles into a ring-shaped structure that underpins virulence complex assembly. PLoS Pathog. 2019 May 9;15(5):e1007761
Other associated proteins
| PFID | Formal Annotation |
|---|---|
| PfEMP1 | PfEMP1 |