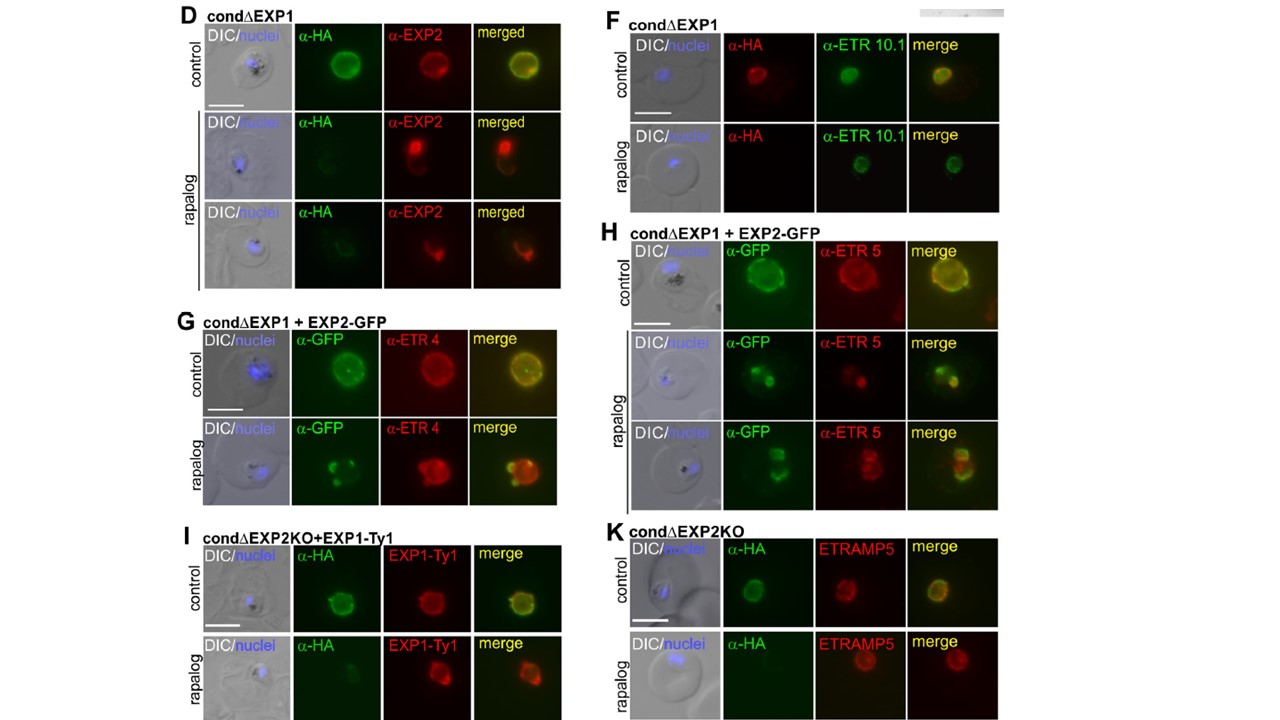
Localization of EXP2 and PVM proteins in ΔEXP1 parasites and interaction analysis. (D). Red, signal around the parasite; black, aberrant distribution of signal. Mean of a total of N = 108 control and N = 135 ΔEXP1 cells derived from 3 biological replicas. (F–H) IFA images of ΔEXP1 (rapalog) and control ring stages (F) or trophozoites (G, H) probed with α-HA (EXP1*-HA) and α-ETRAMP10.1 (F), α-GFP (EXP2-GFP) with α-ETRAMP4 (G), or with α-ETRAMP5 (H). (I) IFA images of ΔEXP2 (rapalog) and control trophozoites expressing EXP1-Ty probed with α-Ty and α-HA to detect EXP1-Ty and EXP2-HA, respectively (K) IFA images of ΔEXP2 (rapalog) and control parasites probed with α-HA and α-ETRAMP5. In (K), α-HA detects full (control) or truncated (rapalog) EXP2-HA. In (A, C, D, F–I, K), scale bar: 5 μm. DAPI, nuclei. Altered distribution of the endogenous EXP2 in ΔEXP1 parasites (D and E). The distribution of other PVM proteins such as early (ETRAMP10.1) or later (ETRAMP4) integral PVM markers showed no apparent differences in ΔEXP1 parasites compared to controls (F and G). In contrast, ETRAMP5 accumulated in small regions of the PVM where it colocalized with the aberrantly distributed EXP2-GFP (H).
Mesén-Ramírez P, Bergmann B, Tran TT, Garten M, Stäcker J, Naranjo-Prado I, Höhn K, Zimmerberg J, Spielmann T. EXP1 is critical for nutrient uptake across the parasitophorous vacuole membrane of malaria parasites. PLoS Biol. 2019 17(9):e3000473
Other associated proteins
| PFID | Formal Annotation |
|---|---|
| PF3D7_0423700 | early transcribed membrane protein 4 |
| PF3D7_0532100 | early transcribed membrane protein 5 |
| PF3D7_1001500 | early transcribed membrane protein 10., ETRAMP10.1 |
| PF3D7_1121600 | parasitophorous vacuole membrane antigen QF 116 exported protein 1 circumsporozoite-related antigen |